CV – Research – Lab members – Publications
 Dr. Ting-Feng Lin
Dr. Ting-Feng Lin
Cell Biology, Neurobiology and Biophysics, Faculty of Science, Utrecht University
Kruytgebouw, room N511
Padualaan 8, 3584 CH Utrecht
The Netherlands
Tel. +31 30 253 2659
e-mail: t.f.lin@uu.nl
Curriculum Vitae
Ting-Feng Lin began his scientific training as a master’s student in the laboratory of Dr. Chih-Yung Tang at National Taiwan University where he investigated biophysical properties of voltage-gated K+ channels (Eag and Erg) and identified a novel role of the N terminus in voltage-dependent inactivation that plays an important role in the cardiac delayed rectifier K+ current and in regulating the intrinsic electrophysiological property of several neuron types.
After His master’s study, he pursued my Ph.D. in the oculomotor laboratory of Dr. Melody Ying-Yu Huang at the University Hospital of Zurich (Neuroscience Center of Zurich, UZH and ETH Zurich), where he discovered and characterized an adaptive oculomotor behavior in larval zebrafish, linking it to human foveal hypoplasia and infantile nystagmus syndrome.
As a postdoc with Dr. Christian Hansel at the University of Chicago, he investigated cerebellar plasticity in intact, awake mice. He first demonstrated that Purkinje cell receptive fields undergo plasticity through both synaptic and intrinsic mechanisms. Furthermore, he showed that predictive parallel fiber ramping activity facilitates LTD in the in vivo cerebellum.
Since September 2025, he started his own lab in the Division of Cell Biology, Neurobiology, and Biophysics, Department of Biology at Utrecht University, where he uses zebrafish to study cellular and circuit neuroscience.
A pdf of a short CV can be found here.
Research summary

Throughout the history of scientific progress, few questions have been as profound, and as mysterious, as how memory is encoded in the brain.
The so-called little brain—the cerebellum—traditionally recognized for its role in motor coordination, is also essential for cognitive functions, owing to its capacity for learning and memory. Impaired cerebellar learning not only disrupts sensorimotor coordination but also contributes to disorders such as autism spectrum disorder (ASD).
My group is dedicated to uncovering the mechanisms of learning and memory, with a particular focus on the cerebellum.
Interactive plasticity mechanisms in the cerebellum
Learning and memory formation require the coordination of multiple plasticity mechanisms—such as synaptic, intrinsic and structural—none of which are sufficient on their own. Although individual mechanisms and their separate effects on neuronal signaling have been well-characterized in vitro, we don’t know how they coordinate, or how that operates in live animals.
Leveraging advanced microscopy in the transparent zebrafish brain, our research aims to decipher these complex interactions at the subcellular level and assess their impact on whole-brain dynamics and behavior. Using the cerebellum as a primary model, we will investigate how diverse mechanisms—each with unique functional, spatial, and temporal characteristics—interact within intricate neuronal structures to regulate downstream signaling. With its evolutionarily conserved architecture, the cerebellum offers a valuable framework to elucidate how plasticity mechanisms interact within neural circuits that are directly relevant to human nervous system function in healthy and diseased conditions.
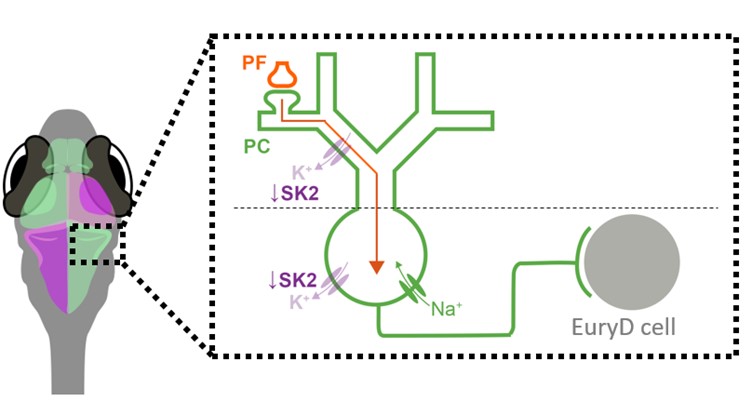
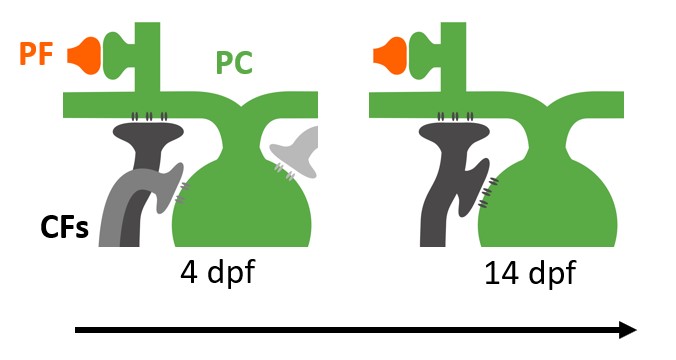
Cerebellar function development
The cerebellum has been implicated in neurodevelopmental disorders such as ASD and schizophrenia. Though often considered opposite ends of a spectrum, these two disorders share a key feature—sensory processing deficits—which has been linked to cerebellar dysfunction. A critical circuit involved in cerebellar sensory processing is the climbing fiber (CF) pathway, which relays instructive signals, such as sensory prediction errors, from the brainstem to the cerebellum. These signals guide synaptic plasticity and contribute to sensory learning.
Proper CF development ensures that sensory prediction errors are accurately assigned to the relevant context, while improper development may cause misattribution of sensory information in neurodevelopmental disorders, such as schizophrenia and ASD. This study will investigate the mechanisms underlying this developmental algorithm by tracking CF function and morphology in larval zebrafish exposed to manipulated sensory environments or optogenetic/chemogenetic neuronal stimulation.
The goal is to determine how sensory experiences shape cerebellar processing during development. Ultimately, these mechanisms will be examined in Grid2 knockout—a shared genetic factor in ASD and schizophrenia characterized by cerebellum-specific pathology. By integrating advanced imaging, optogenetic, and chemogenetic approaches, this study will elucidate fundamental mechanisms of cerebellar circuit maturation in healthy and diseased conditions.
Lab members
| PhD students: | |
| Mick de Koning | |
| Master students: | |
| Deirbhile Cruise | |
| Joanne van Krimpen |
Publications
For a complete list of publications please visit the google scholar page.
2024
Lin TF, Grasselli G, Hansel C*. Cellular Mechanisms Underlying Cerebellar Learning. In Byrne JH (Ed.). Learning and Memory: A Comprehensive Reference, 3rd Edition. Elsevier Science. 2024. (Book chapter; hardcopy in press)
Lin TF, Busch, S.E., Hansel, C*. Intrinsic and synaptic determinants of receptive field plasticity in Purkinje cells of the mouse cerebellum. Nature Communications. 2024.
2023
Lin TF*, Huang MY*. A quantitative approach to study the adaptation of rhythmic eye movements and the resulting tonic eye deviation in larval zebrafish. J Neurosci Res. 2023.
2022
Lin TF*, Mohammadi M, Cullen KE, Chacron MJ, Huang MY*. Optokinetic set-point adaptation functions as an internal dynamic calibration mechanism for oculomotor disequilibrium. iScience. 2022.
2019
Lin TF, Mohammadi M, Fathalla AM, Pul D, Lüthi D, Romano F, Straumann D, Cullen KE, Chacron MJ, Huang MY*. Negative optokinetic afternystagmus in larval zebrafish demonstrates set-point adaptation. Sci Rep. 2019.
2018
Lin TF, Gerth-Kahlert C, Hanson JVM, Straumann D, Huang MY*. Spontaneous nystagmus in the dark in an infantile nystagmus patient may represent negative optokinetic afternystagmus. Front Neurol. 2018.
2016
Hsu PH, Chiu YC, Lin TF, Jeng CJ*. Ca2+-binding protein centrin 4 is a novel binding partner of rat Eag1 K+ channels. FEBS Open Bio. 2016.
2014
Lin TF, Jow GM, Fang HY, Fu SJ, Wu HH, Chiu MM, Jeng CJ*. The eag domain regulates the voltage-dependent inactivation of rat eag1 k+ channels. PLoS One. 2014.
Lin TF, Lin IW, Chen SC, Wu HH, Yang CS, Fang HY, Chiu MM, Jeng CJ*. The subfamily-specific assembly of Eag and Erg K+ channels is determined by both the amino and the carboxyl recognition domains. J Biol Chem. 2014.

