CV – Research – Lab members – Publications
 Dr. Agathe Chaigne
Dr. Agathe Chaigne
Cell Biology, Neurobiology and Biophysics,
Faculty of Science, Utrecht University
Kruytgebouw, room N501
Padualaan 8, 3584 CH Utrecht, The Netherlands
a.e.d.chaigne@uu.nl
@agathe.chaigne.bsky.social
Curriculum Vitae
Agathe Chaigne studied cell biology and developmental biology in Ecole Normale Supérieure and Université Pierre et Marie Curie, in Paris, France. She obtained a teaching degree in 2010, her masters in 2011 and her PhD in 2014 studying the role of cortex tension in spindle positioning of the mouse oocyte, under the supervision of Marie-Hélène Verlhac and Marie-Emilie Terret. She did her postdoc in the lab of Ewa Paluch in University College London and Cambridge University, in collaboration with Kevin Chalut and Edouard Hannezo as an EMBO fellow then a sir Henry Wellcome postdoctoral fellow. During that time, she investigated the role of cell division in cell fate transitions using mouse embryonic stem cells as a model system. She started her own lab at the department of Cell Biology, Neurobiology and Biophysics in January 2022 and was awarded a NWO-VIDI grant in the same year.
Research summary
 Cell division is crucial for development: it allows an increase in the number of cells, while also being important for cell fate transitions. Indeed, cell division can produce cells that are different from the mother cells, or different from each other in the case of asymmetric division.
Cell division is crucial for development: it allows an increase in the number of cells, while also being important for cell fate transitions. Indeed, cell division can produce cells that are different from the mother cells, or different from each other in the case of asymmetric division.
We aim at understanding how the fundamental mechanisms of cell division can control fate transitions, with a developmental and evolutionary perspective. We are particularly interested in the last step of cell division, abscission, which is the final cut between the sister cells. While the textbook picture of cell division describes abscission following shortly cytokinesis, some cells do not perform abscission but instead remain connected by cytoplasmic bridges for extended periods of time. We are interested in the mechanisms of formation, maintenance, and roles of the cytoplasmic bridges (Figures created using Biorender.com).
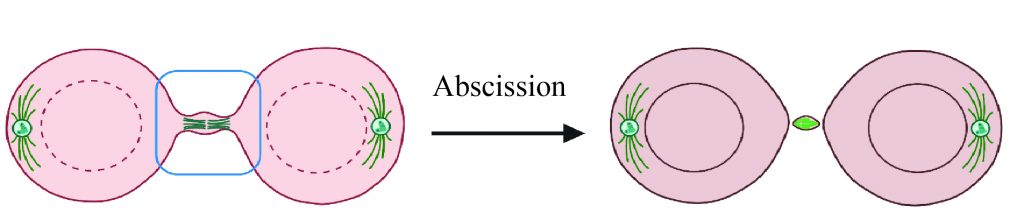
Using live-cell imaging, comparative approaches, proteomics, and manipulation of abscission, we are currently focusing on two specific questions:
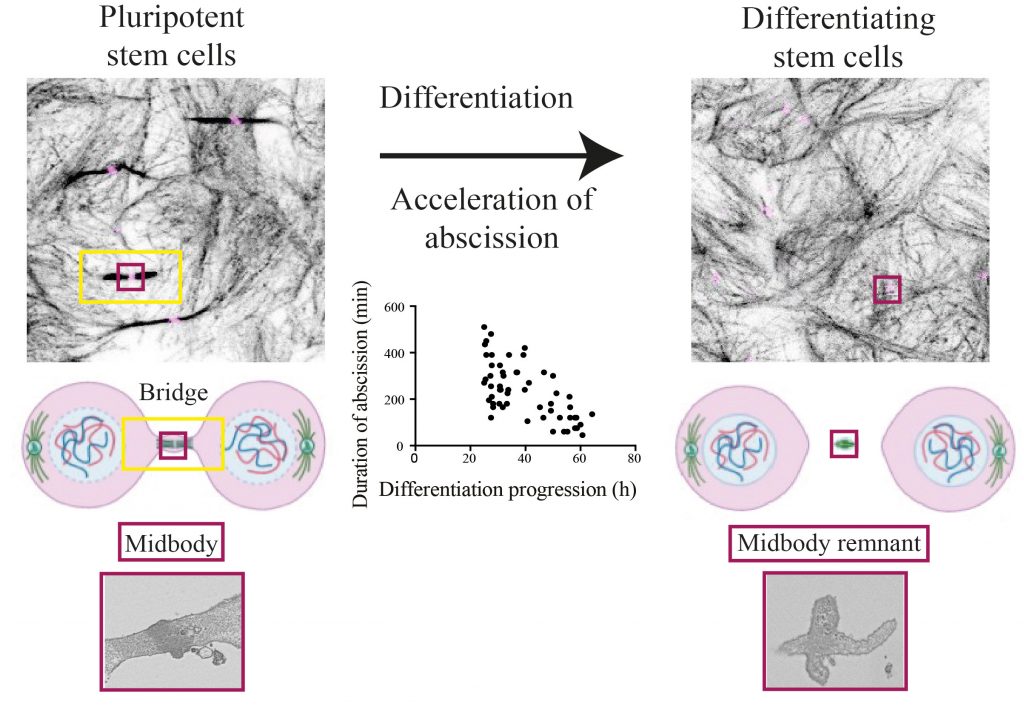
- How are bridges maintained and what are their roles in early development? To answer this question, we are using mouse embryonic stem cells as a model system. Pluripotent stem cells maintain long-lived bridges and dismantling of bridges through accelerated abscission is crucial to exit from naïve pluripotency.
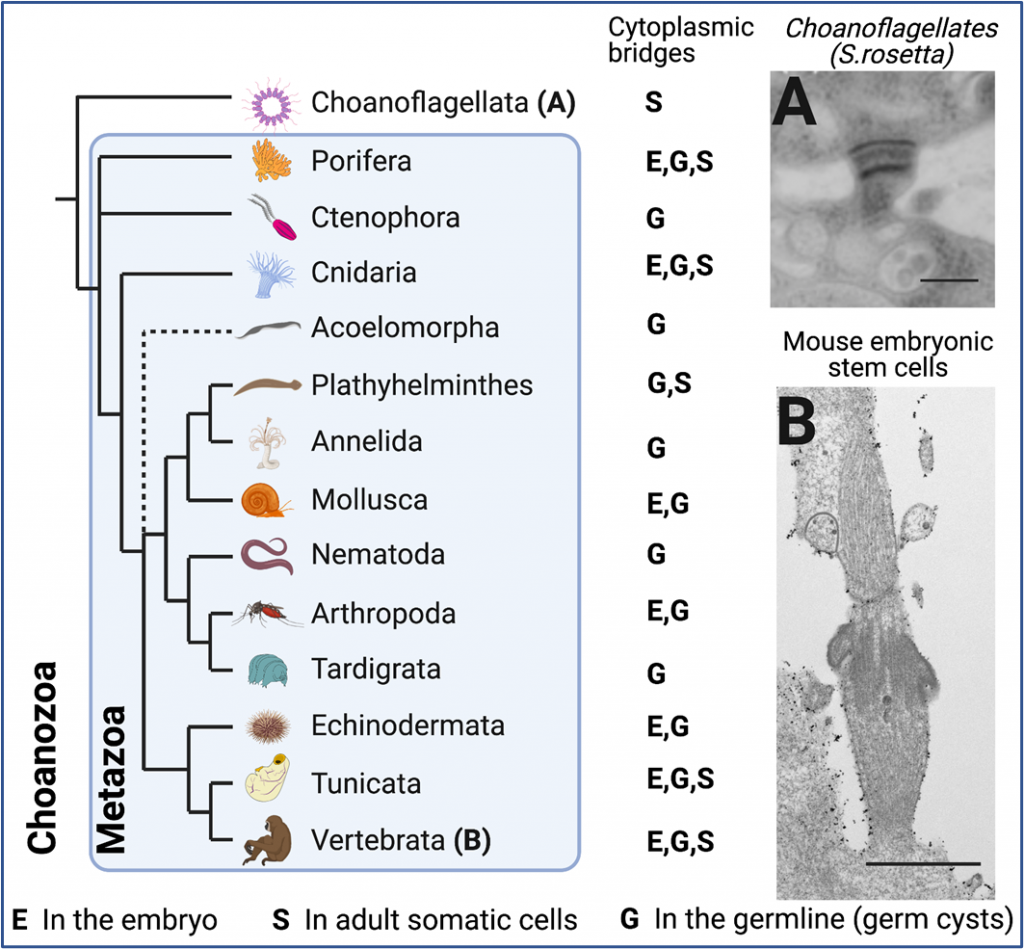
- What role did bridges play in the evolution of animal multicellularity? To address this question, we plan on comparing bridges in different species, in particular in choanoflagellates, the closest living relatives to animals.
We are not currently recruiting for specific projects, but we are always looking for motivated candidates. Please email Agathe if interested.
Lab members
| Postdoc: | |
| My Anh Truong | m.a.truong@uu.nl |
| PhD students: | |
| Snježana Kodba | s.kodba@uu.nl |
| Amber Öztop | a.d.h.oztop@uu.nl |
| Nils Dormanns | n.p.dormanns@uu.nl |
| Technician: | |
| Erika Timmers | e.t.timmers@uu.nl |
Publications
Articles
2024
Snježana Kodba, Amber Öztop, Eri van Berkum, Malina K. Iwanski, Wilco Nijenhuis, Lukas C. Kapitein, Agathe Chaigne, Aurora B controls microtubule stability to regulate abscission dynamics in stem cells. BioRxiv 2024 March, doi: https://doi.org/10.1101/2024.03.06.583686
2023
Smith MB, Sparks H, Almagro J, Chaigne A, Behrens A, Dunsby C, Salbreux G. Active mesh and neural network pipeline for cell aggregate segmentation. Biophys J. 2023 May 2;122(9):1586-1599.
2021
Chaigne, A., Smith, M. B., Lopez Cavestany, R., Hannezo, E., Chalut, K. J. and Paluch, E. K. (2021). Three-dimensional geometry controls division symmetry in stem cell colonies. Journal of Cell Science 134, jcs255018.
2020
Chaigne, A., Labouesse, C., White, I.J., Agnew, M., Hannezo, E., Chalut, K.J., and Paluch, E.K. (2020). Abscission Couples Cell Division to Embryonic Stem Cell Fate. Developmental Cell.
Dimitracopoulos, A.*, Srivastava, P*, Chaigne, A., Win, Z., Shlomovitz, R., Lancaster, O.M., Le Berre, M., Piel, M., Salbreux, G., Baum, B. Mechanochemical crosstalk produces intrinsic patterning of the cell cortex to orient the mitotic spindle. Current Biology.
Bennabi, I., Crozet, F., Nikalayevich, E., Chaigne, A., Letort, G., Manil-Ségalen, M., Campillo, C., Cadart, C., Othmani, A., Attia, R., et al. (2020). Artificially decreasing cortical tension generates aneuploidy in mouse oocytes. Nature Communications 11.
2016
Smith, M. B., Chaigne, A. & Paluch, E. K. (2016). An active contour ImageJ plugin to monitor daughter cell size in 3D during cytokinesis. In Methods in Cell Biology.
Chaigne, A., Campillo, C., Voituriez, R., Gov, N.S., Sykes, C., Verlhac, M.-H., and Terret, M.-E. (2016). F-actin mechanics control spindle centring in the mouse zygote. Nat. Commun. 7, 10253.
2015
Chaigne, A., Campillo, C., Gov, N.S., Voituriez, R., Sykes, C., Verlhac, M.H., and Terret, M.E. (2015). A narrow window of cortical tension guides asymmetric spindle positioning in the mouse oocyte. Nat. Commun. 6, 6027.
2013
Chaigne, A., Campillo, C., Gov, N.S.*, Voituriez, R.*, Azoury, J., Umaña-Diaz, C., Almonacid, M., Queguiner, I., Nassoy, P., Sykes, C., Verlhac, M.-H., Terret, M.-E., 2013. A soft cortex is essential for asymmetric spindle positioning in mouse oocytes. Nat. Cell Biol. 15, 958–966.
Reviews & Protocols
Öztop A, Chaigne A. Molecular and mechanical mechanisms of animal cell abscission. FEBS Lett. 2024 Sep 26. doi: 10.1002/1873-3468.15015. Epub ahead of print. PMID: 39324548.
Kodba S, Chaigne A. Delayed abscission in animal cells – from development to defects. J Cell Sci. 2023 Jul 1;136(13):jcs260520. doi: 10.1242/jcs.260520.
Chaigne A, Brunet T. Incomplete abscission and cytoplasmic bridges in the evolution of eukaryotic multicellularity. Curr Biol. 2022 Apr 25;32(8):R385-R397. doi: 10.1016/j.cub.2022.03.021.
Kodba S, Chaigne A. A quick, cheap, and reliable protocol for immunofluorescence of pluripotent and differentiating mouse embryonic stem cells in 2D and 3D colonies. STAR Protoc. 2023 Jan 20;4(1):102000. doi: 10.1016/j.xpro.2022.102000.
Carla Mulas, Agathe Chaigne, Austin Smith, Kevin J. Chalut; Cell state transitions: definitions and challenges. Development 15 October 2021; 148 (20): dev199950. doi: https://doi.org/10.1242/dev.199950
Chaigne, A. In Vitro Fertilization of Mouse Oocyte. Methods Mol. Biol. 1818, 37–42 (2018).
Chaigne, A., Terret, M.-E. & Verlhac, M.-H. Asymmetries and Symmetries in the Mouse Oocyte and Zygote. in Asymmetric Cell Division in Development, Differentiation and Cancer (eds. Tassan, J.-P. & Kubiak, J. Z.) 61, 285–299 (Springer International Publishing, 2017).
Chaigne, A., Verlhac, M.-H., Terret, M.-E., 2014. Ramollir le cortex: Un prérequis à l’asymétrie de la division ovocytaire. médecine/sciences 30, 18–21.
Terret, M.-E., Chaigne, A., Verlhac, M.-H., 2013. Mouse oocyte, a paradigm of cancer cell. Cell Cycle 12, 0–6.
Chaigne, A., Verlhac, M.-H. and Terret, M.-E. 2012. Spindle positioning in mammalian oocytes. Experimental Cell Research 318, 1442–1447.
