Kah Wai Yau, Sam F.B. van Beuningen, Inês Cunha-Ferreira, Bas M.C. Cloin, Eljo Y. van Battum, Lena Will, Philipp Schätzle, Roderick P. Tas, Jaap van Krugten, Eugene A. Katrukha, Kai Jiang, Phebe S. Wulf, Marina Mikhaylova, Martin Harterink, R. Jeroen Pasterkamp, Anna Akhmanova, Lukas C. Kapitein, and Casper C. Hoogenraad
http://dx.doi.org/10.1016/j.neuron.2014.04.019
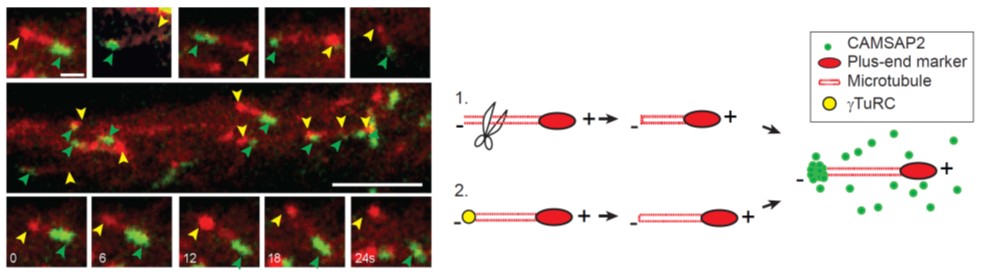
In neurons, most microtubules are not associated with a central microtubule-organizing center (MTOC), and therefore, both the minus and plus-ends of these noncentrosomal microtubules are found throughout the cell. Microtubule plus-ends are well established as dynamic regulatory sites in numerous processes, but the role of microtubule minus-ends has remained poorly understood. Using live-cell imaging, high-resolution microscopy, and laser-based microsurgery techniques, we show that the CAMSAP/Nezha/Patronin family protein CAMSAP2 specifically localizes to noncentrosomal microtubule minus-ends and is required for proper microtubule organization in neurons. CAMSAP2 stabilizes noncentrosomal microtubules and is required for neuronal polarity, axon specification, and dendritic branch formation in vitro and in vivo. Furthermore, we found that noncentrosomal microtubules in dendrites are largely generated by γ-Tubulin dependent nucleation. We propose a two-step model in which γ-Tubulin initiates the formation of noncentrosomal microtubules and CAMSAP2 stabilizes the free microtubule minus-ends in order to control neuronal polarity and development.





 Anna Akhmanova:
Cellular Dynamics
Anna Akhmanova:
Cellular Dynamics Lukas Kapitein: Biophysics
Lukas Kapitein: Biophysics Florian Berger:
Theoretical Biophysics
Florian Berger:
Theoretical Biophysics Agathe Chaigne:
Cell division dynamics
Agathe Chaigne:
Cell division dynamics Ginny Farías:
Neuronal Organelle Dynamics
Ginny Farías:
Neuronal Organelle Dynamics Martin Harterink:
C. elegans neurobiology
Martin Harterink:
C. elegans neurobiology Casper Hoogenraad: Molecular Neuroscience
Casper Hoogenraad: Molecular Neuroscience Maarten Kole:
Axonal Signalling
Maarten Kole:
Axonal Signalling Sabrina Oliveira:
Molecular Targeted Therapies
Sabrina Oliveira:
Molecular Targeted Therapies Ihor Smal: Image Analysis, Smart Microscopy and AI
Ihor Smal: Image Analysis, Smart Microscopy and AI Frederik Verweij:
Extracellular Vesicle Biology
Frederik Verweij:
Extracellular Vesicle Biology